AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Sodium element model1/31/2024  The metallic bond is not fully broken until the metal boils. 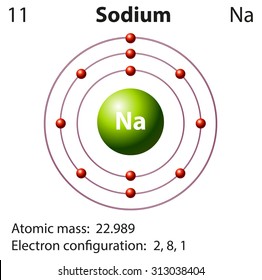
In a molten metal, the metallic bond is still present, although the ordered structure has been broken down. The "Sea of Electrons" theory stands today only as an oversimplified model of how metallic bonding works. However, these observations are only qualitative, and not quantitative, so they cannot be tested. Electrons on the surface can bounce back light at the same frequency that the light hits the surface, therefore the metal appears to be shiny. Luster: The free electrons can absorb photons in the "sea," so metals are opaque-looking.Heat capacity: This is explained by the ability of free electrons to move about the solid.Thus if we hit a metal with a hammer, the crystals do not shatter, but merely change their shape, This is very different from the behavior of ionic crystals. The final situation is much the same as the initial. Global positioning system (GPS) signals must be accurate to within a billionth of a second per day, which is equivalent to gaining or losing no more than one second in 1,400,000 years.\): Malleability of metals originate from each of moving layer of atoms with respect to each other. Telecommunications systems, such as cell phones, depend on timing signals that are accurate to within a millionth of a second per day, as are the devices that control the US power grid. In contemporary applications, electron transitions are used in timekeeping that needs to be exact. The Paschen, Brackett, and Pfund series of lines are due to transitions from higher-energy orbits to orbits with n = 3, 4, and 5, respectively these transitions release substantially less energy, corresponding to infrared radiation. 
These transitions are shown schematically in Figure 2.3.4įigure 2.3.4 Electron Transitions Responsible for the Various Series of Lines Observed in the Emission Spectrum of Hydrogen The Lyman series of lines is due to transitions from higher-energy orbits to the lowest-energy orbit ( n = 1) these transitions release a great deal of energy, corresponding to radiation in the ultraviolet portion of the electromagnetic spectrum. Other families of lines are produced by transitions from excited states with n > 1 to the orbit with n = 1 or to orbits with n ≥ 3. Consequently, the n = 3 to n = 2 transition is the most intense line, producing the characteristic red color of a hydrogen discharge (part (a) in Figure 2.3.1 ). At the temperature in the gas discharge tube, more atoms are in the n = 3 than the n ≥ 4 levels. Because a sample of hydrogen contains a large number of atoms, the intensity of the various lines in a line spectrum depends on the number of atoms in each excited state. Name: Sodium Atomic Number: 11 Element Symbol: Na Group: 1 Period: 3 Block: s Element Family: alkali metal Atomic Mass: 22.989 769 28(2) Electron Configuration: Ne3s 1 (shorthand) or 1s 2 2s 2 2p 6 3s 1 (full) Discovery: Sir Humphry Davy in 1807 Davy isolated sodium from sodium hydroxide using electrolysis. The n = 3 to n = 2 transition gives rise to the line at 656 nm (red), the n = 4 to n = 2 transition to the line at 486 nm (green), the n = 5 to n = 2 transition to the line at 434 nm (blue), and the n = 6 to n = 2 transition to the line at 410 nm (violet). Thus the hydrogen atoms in the sample have absorbed energy from the electrical discharge and decayed from a higher-energy excited state ( n > 2) to a lower-energy state ( n = 2) by emitting a photon of electromagnetic radiation whose energy corresponds exactly to the difference in energy between the two states (part (a) in Figure 2.11 ). As shown in part (b) in Figure 2.11, the lines in this series correspond to transitions from higher-energy orbits ( n > 2) to the second orbit ( n = 2). We can now understand the physical basis for the Balmer series of lines in the emission spectrum of hydrogen (part (b) in Figure 2.9 ). Bohr calculated the value of \(\Re\) from fundamental constants such as the charge and mass of the electron and Planck's constate and obtained a value of 1.0974 × 10 7 m −1, the same number Rydberg had obtained by analyzing the emission spectra. The negative sign in Equation 2.11 and Equation 2.12 indicates that energy is released as the electron moves from orbit n 2 to orbit n 1 because orbit n 2 is at a higher energy than orbit n 1. \)Įxcept for the negative sign, this is the same equation that Rydberg obtained experimentally. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
